|
NOVIDADES
Hydrogen is a clean fuel that can be burned in a fuel cell to produce energy with minimal impact to the environment. One method to produce hydrogen is by using electricity to spilt water molecules, in the presence of a catalyst. NUS researchers have developed a way to create stable and selective catalysts which can be used for efficient hydrogen production (Nature Communications, "Interface confined hydrogen evolution reaction in zero valent metal-intercalated molybdenum disulfide"). Their method encapsulate noble metal nanoparticles in quasi-2D materials by a simple in-situ reduction method. This is like putting the ingredients between pieces of bread in a sandwich. This new method is an easier way to produce this structure, avoiding the tedious exfoliation process of 2D nanosheets. The confined space within the quasi-2D layers provides a well controlled environment for catalysis to take place. It also prevents larger sized pollutants or neutral molecules from affecting the catalytic process. In their tests, the catalysts show excellent activity and long-term stability when they are used for hydrogen production. 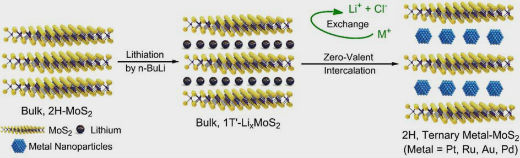 This figure shows the encapsulation of noble metal nanoparticles in MoS2 by an in-situ reduction strategy. Image: NUS Interface confined reactions, which can modulate the bonding of reactants with catalytic centres and influence the rate of the mass transport from bulk solution, have emerged as a viable strategy for achieving highly stable and selective catalysis. However, the confinement of nanoparticles in 2D, layered materials is challenging due to the strong van der Waals force between adjacent nanosheets. Conventional methods relying on the diffusion of ion precursors by capillary force are not feasible to achieve this goal. Prof LOH Kian Ping, together with his Ph.D. students CHEN Zhongxin, LENG Kai, ZHAO Xiaoxu, from the Department of Chemistry, NUS, employed an ingenious strategy based on the in-situ reduction of ion precursors to introduce nanoparticles into inner spaces of the host material. The growth of nanoparticles within a confined space results in a smaller particle size with improved catalytic performance. This research breakthrough is achieved in collaboration with electrochemist, Prof YEO Boon Siang from the Department of Chemistry, NUS. Prof Yeo investigated the anisotropic diffusion kinetics of reactants to explain the excellent long-term stability of these catalysts. Unlike other research work on 2D catalysts, this work does not involve the exfoliation of 2D nanosheets, which is a complicated process. Instead, the researchers took advantage of highly reductive, lithiated host materials (LixMoS2) to react with ion precursors. This provides a strong driving force to overcome van der Waals interactions and transforms the quasi-2D materials into a unique, MoS2 | noble metals | MoS2 sandwich structure (Figure). The researchers further demonstrated industrial scalability by fabricating and testing a 25 cm2 catalyst-loaded water splitting membrane. This is unprecedented in current 2D research, which are often limited by the size of the exfoliated flakes and difficulty in spin-coating a continuous film. The synergetic host–guest interaction allows for ultra-stable, long-term operation of the catalyst for hydrogen production. It also has a reduced metal loading compared with commercially available catalyst. This new concept of confinement using 2D materials can potentially be applied to many other catalytic reactions involving energy-related applications. National University of Singapore. Posted: jul 21, 2017. |
|||||||||||||||||||||||||