|
NOVIDADES
The term personalized medicine describes medical decisions that are tailored to individual patients or specific groups of patients. This type of approach has the potential to maximize efficacy by providing each patient with optimal care while avoiding ineffective remedies. Vaccines, both against bacteria and tumors, are a clinical modality that could benefit significantly from personalization. Effective vaccination strategies could help to address many challenging disease conditions, but current vaccines are limited by factors such as a lack of potency and antigenic breadth. Recently, researchers have turned toward the use of biomimetic nanotechnology as a means of addressing these hurdles. A just published progress report in Advanced Materials ("Biomimetic Nanotechnology toward Personalized Vaccines") discusses recent progress in the development of biomimetic nanovaccines for antibacterial and anticancer applications, with an emphasis on their potential for personalized medicine. The authors, from the Department of NanoEngineering, Chemical Engineering Program, and Moores Cancer Center at the University of California San Diego, begin with a basic immunology overview and discuss the principles governing effective vaccine design. They then discuss in detail the advantages of incorporating nanotechnology in the engineering of vaccines and the current state of nanovaccines. Finally, they review recent developments in nanovaccines for antibacterial and anticancer therapy, with a special focus on biomimetic platforms that have future implications for personalized medicine. 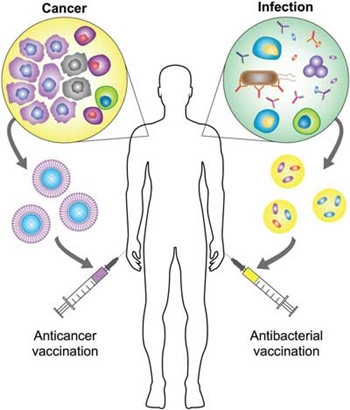 Personalized biomimetic nanovaccines. For anticancer vaccination, antigenic material can be collected directly from a patient’s resected tumor, formulated into a biomimetic nanoparticle, and then administered back into the patient to promote tumor-specific immunity. For antibacterial vaccination, strain-specific virulence factors or membrane can be immobilized onto nanoparticle substrates, and the resulting complexes can be used to vaccinate patients with an identified risk against the associated pathogen. Reprinted with permission by Wiley-VCH Verlag
To achieve codelivery, the adjuvant can either be encapsulated into the nanoparticle core, functionalized onto the surface, or the nanoparticle material itself can serve as the stimulus. Another advantage of nanoparticle-based vaccines is the enhanced bioavailability of the payloads. By encapsulating or conjugating antigen and adjuvant to a nanocarrier, the materials can more effectively be protected from host interactions during transport. In addition, nanoparticulate delivery can also prevent systemic toxicities that are often associated with the administration of adjuvants in their free form. Thus, by shielding their payloads from the surrounding environment, nanoparticles can concurrently protect the host from nonspecific biologicals interactions, which can cause unintended side effects. The unique size range of nanoparticles is another factor that can enable improved delivery of vaccine components. The nanoscale dimensions of nanocarriers allow for more efficient lymphatic drainage into the lymphoid organs where antigen uptake and processing can occur. Nanoparticles can also be designed for efficient delivery into the cell cytosol – the liquid found inside cells – which has major implications for improving vaccine performance. For cancer vaccine applications, effective cytosolic delivery can benefit both antigen and adjuvant, leading to simultaneous enhancement of antigen presentation and improvement of immune stimulation. Advantages of nanovaccines have been similarly utilized for managing bacterial infections. Nanotechnology can confer several unique advantages when it comes to the engineering of vaccine formulations. Careful manipulation of nanoparticle parameters and conscious design choices can significantly improve potency compared with traditional vaccine delivery systems. 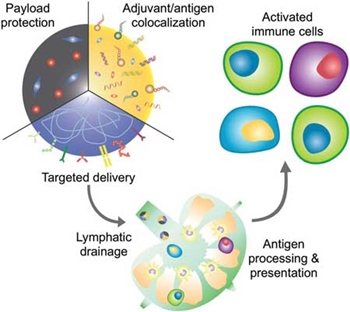 Reprinted with permission by Wiley-VCH Verlag
This is where biomimetics comes in i.e., the imitation of the models, systems, and elements of nature for the purpose of designing technology. One way in which biomimetic design can enhance the utility of nanoparticle technology is by enabling targeted delivery through the use of natural ligands. In addition to targeting interactions, biomimetic functionalization of nanoparticles can also be used to modulate the activity of biological targets. This can be particularly useful for biodetoxification, where therapies are designed to neutralize the activity of toxic molecules that pose a threat to human health. Nanoparticles functionalized with natural cell membrane have proven to be useful for a number of biomedical applications. For example, by camouflaging nanoparticle surfaces with red blood cell membrane, it has been demonstrated that the immune system will recognize the resulting nanoparticles as self rather than foreign.
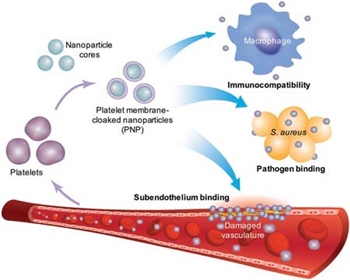 Platelet membrane–coated nanoparticles. Platelet membrane is derived from whole platelets by a repeated freeze–thaw process. The purified membranes can then be fused with a nanoparticulate core, enabling natural targeting affinity toward pathogens and damaged vasculature. Reprinted with permission by Springer Nature
The emergence of 'superbug' pathogens that are resistant to most if not all antibiotics, has led to increased research efforts for developing innovative strategies that are focused on prevention rather than treatment. This is an area in which biomimetic nanotechnology can provide significant benefits, and increasing attention has been placed on the use of extracellular vesicles and nanotoxoids as antibacterial vaccines. Both of these biomimetic systems are amenable to personalization, where formulations can be facilely tailored to address any number of individual bacterial strains or fabricated on-demand for specific patient populations. Anticancer therapy is another area in which there is a high demand for personalized medicine. The pathogenesis of cancer is extremely complex, and the disease is inherently hard to treat given that malignant cells are derived from mutated versions of one’s own healthy cells. With many disease-causing factors involved, cancer varies greatly from patient to patient. Recently, researchers have engineered different biomimetic platforms, including cell-derived nanovesicles, virus-like particles, artificial antigen-presenting cells, and cell membrane–coated nanoparticles, for use as anticancer vaccines. Many of these have the potential to develop into personalized therapies that can ultimately help to overcome tumor heterogeneity. The authors conclude that vaccination has become an increasingly attractive option for disease management given its ease of use, broad applicability, and ability to generate long-term protection. Biomimetic nanovaccines can be inherently multiantigenic and immunostimulatory, as is the case with outer membrane vesicles (OMV) and OMV-coated nanoparticles. A newly emerging approach leverages cell membrane–coated nanoparticles for neutralizing and delivering bacterial toxins for antivirulence vaccination. By vaccinating against the tools that pathogens utilize for survival, this strategy can effectively prevent colonization while limiting the direct selective pressure that drives antibiotic resistance. As the membrane coating can be easily interchanged and toxin mixtures from different bacteria can be used to supply the antigenic material, a countless number of nanotoxoid formulations can be developed, which may ultimately enable more personalized vaccines that can be tailored to individual patient populations with specific risk profiles. By Michael Berger – Michael is author of three books by the Royal Society of Chemistry: Nano-Society: Pushing the Boundaries of Technology, Nanotechnology: The Future is Tiny, and Nanoengineering: The Skills and Tools Making Technology Invisible NW. Posted: Apr 03, 2020. |
|||||||||||||||||||||||||