|
NOVIDADES
The demand for low-cost water purification technologies has become a pressing issue due to the rapid population growth in the world. At the turn of the last millennium, we already had over 1 billion people lacking access to safe drinking water and basic sanitation, and nearly 4,000 children under the age of five dying each day, the term clean water crisis today seems like an understatement. (1) In this article, we focus on one possible pathway to tackle the above challenges by using the abundant, renewable and inexpensive natural plant biomass as a sustainable source to extract nanoscale materials (nanocellulose) for water purification. Nanocellulose can be used as a valuable sorptive material, comparable to activated carbon or carbon nanomaterial, to remove contaminants (2). 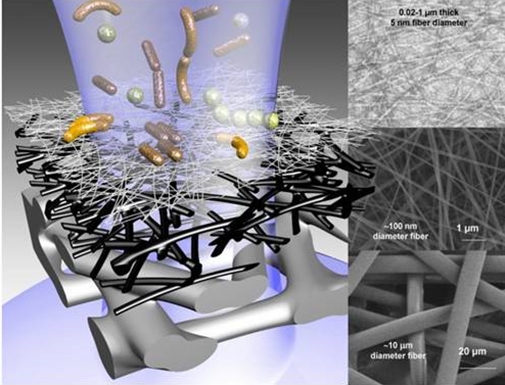 Figure 1: A three-layered nanocellulose filter with hierarchical fibre diameters from microns to nanometers. Credit: OpenAcessGov
Cellulose is the most abundant natural polymer on earth. Its annual production is in the order of 7.5 x 1010 ton, which corresponds to an annual carbon production of 30 x 109 ton. This carbon production could be compared with the total carbon emissions from fossil fuel consumption, which amounts to 9 x 109 ton. (4) Cellulose is produced by higher plants, a wide range of varying bacteria, algae and fungi, and certain animals such as tunicates. Both woody and non-woody plant fibres can be used to extract nanocellulose. A general consensus is that non-woody plants usually have a lower lignin content than woody plants, which make these non-woody plant fibres easier to delignify thus facilitating the nanocellulose extraction process. The current chemistries for nanocellulose extraction have mainly been developed to deal with woody plants. For lower value non-woody plants, such as agricultural residues, the major focus today is the production of biofuels. We argue that as the logistics of collection, transportation and decortication for biofuel production are being worked out to deal with agricultural residues, the exploration of new and simple chemistries that can extract nanocellulose from non-woody plants in a more cost-effective manner than the existing approaches using wooden plants can definitely enable us to develop low-cost nanocellulose processes to upcycle vastly underutilised non-wooden biomass especially for water purification. Often, the agricultural residues are utilised mainly as a burning source for waste removal or energy generation, which cause undesired air pollution. During the past decades, there has been a rapid growth of research and commercial interests towards the development of nanocellulose materials. The production methods can be divided into two paths: the bottom-up approach that involves the use of bacteria to convert sugars into nanocellulose (bacterial nanocellulose); the top-down approach that involves the use of varying enzymatic/chemical /mechanical processes to break down fibres to their elementary microfibrillar or their aggregate forms. The use of chemical pre-treatments to facilitate the defibrillation of lignocellulose fibres into CNF can be categorised into two classes. (1) Electrostatically induced swelling by the introduction of charged groups onto cellulose chains, which can be accomplished in either the pulping or bleaching step or by subjecting cellulose fibres to oxidative treatment. (2) Mild acid or enzymatic hydrolysis of cellulose fibres. (5) Both processes can decrease the cell wall cohesion and, hence, reduce the energy consumption during defibrillation of cellulose fibres. It is known that the cell walls in pulped wood fibres have naturally occurring charged groups, such as carboxyl groups. These charged groups can facilitate the swelling and decrease the cell wall cohesion in cellulose fibres. The charge content can be significantly increased through several oxidation and chemical modification procedures. The high charge content can drastically decrease the energy consumption during defibrillation of cellulose fibres. Recently, a simple nitro-oxidation method has been developed in our laboratory to prepare CNF directly from raw biomass (jute, spinifex grass and bamboo) using only nitric acid-sodium nitrite mixtures. (6) In this method, the lignin component is depolymerized into soluble products by the presence of nitrogen oxide species (produced by the reaction of nitric acid and sodium nitrite) and the hemicellulose component is broken down by nitric acid. In addition, the generation of nitroxonium ions can selectively oxidize the primary hydroxyl groups of anhydroglucose units of cellulose to carboxyl groups. As a result, the nitro-oxidation method significantly reduces the need for multiple chemicals and the consumption of electric energy and water for producing CNF. Also, the effluent (spent liquor) from this method could be neutralised using the base to produce nitrogen-rich salts as plant fertilisers to avoid expensive recovery operations. (7) R. Sharma, S. K. Sharma, T. Lindström, B. S. Hsiao, Advanced Sustainable Systems 2020, 4 (5), 1900114. A. Boden, G. Marland, R. J. Andres, Global, regional and national fossil-fuel carbon dioxide emissions, U.S. Department of Energy, Oak Ridge, TN, 2010. Pääkkö, M. Ankerfors, H. Kosonen, A. Nykänen, S. Ahola, M. Österberg, J. Ruokolainen, J. Laine, P. T. Larsson, O. Ikkala, T. Lindström, Biomacromolecules 2007, 8, 1934. R. Sharma, R. Joshi, S. K. Sharma, B. S. Hsiao, Biomacromolecules 2017, 18, 2333. I. Brink, Tappi 1961, 44, 256. OpenAcessGov. Posted, September 24, 2020.
|
|||||||||||||||||||||||||