|
NOVIDADES
Protein structures obtained in part at the Advanced Light Source (ALS) helped researchers to increase the potency of simplified antibodies (nanobodies) designed to neutralize SARS-CoV-2. Stable enough to be used in inhalers or nasal sprays, the nanobodies offer a new option, aside from injected vaccines, for COVID-19 prevention and treatment. The findings have been published in Science ("An ultrapotent synthetic nanobody neutralizes SARS-CoV-2 by stabilizing inactive Spike"). 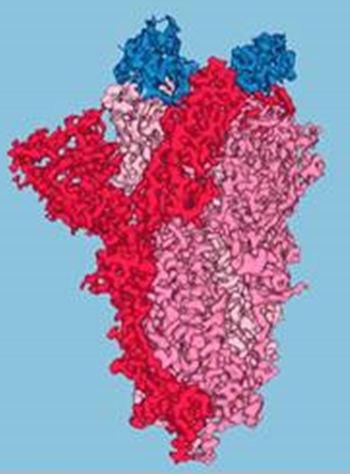 Three nanobodies (blue) are shown bound to the tip of the SARS-CoV-2 spike protein (red/pink). The nanobodies were engineered to be extremely effective at blocking the virus from entering a host cell. Image: UCSF
In the work reported here, researchers from the University of California San Francisco (UCSF) approached the problem through the production of simplified antibodies (“nanobodies”) engineered to be highly effective at blocking SARS-CoV-2, the virus that causes COVID-19. Because nanobodies are extremely stable, they can be aerosolized, stored at room temperature, and self-administered as needed, directly to affected nasal or lung tissues using nasal sprays or inhalers. At UCSF, the researchers sifted through over two billion synthetic nanobody sequences available in databases and identified a few most likely to bind to and neutralize the SARS-CoV-2 spike protein—the distinctive protrusions on the coronavirus surface that facilitate entry to a host cell. Three nanobodies in particular (Nb3, Nb6, and Nb11) were selected for further characterization and study. In cases where cryo-EM provided inconclusive data, the researchers relied on a relatively new technique called x-ray footprinting at ALS Beamline 3.2.1. This technique provides structural information similar to crystallography, but is done with molecules in solution and doesn’t require protein crystals.  T(a) Crystallography data from the ALS show atomic-level details of key Nb6 regions. (b) X-ray footprinting experiments performed at the ALS revealed a cluster of residues in the spike’s N-terminal domain, where it likely interacts with Nb3. All combined, the structural work gave the researchers enough information to optimize the potency of the nanobodies, Nb6 in particular, through strategies such as the linking of two or three nanobodies together (creating “bivalent” and “trivalent” versions that attach to the spike at multiple points) and the targeted replacement of amino acids—mutations that preliminary screens indicated would increase nanobody affinity for the spike. The researchers are currently moving forward to the animal testing phase for these synthetic nanobodies, as their combined stability, potency, and multiple modes of action provide a distinctive potential prophylactic and therapeutic approach to limiting the continued toll of the COVID-19 pandemic. Berkeley Lab. Posted: Jan 26, 2021.
|
|||||||||||||||||||||||||