|
NOVIDADES
Virginia Tech scientists have developed a new cancer drug that uses gold nanoparticles created by the biotech firm CytImmune Sciences to deliver paclitaxel — a commonly used chemotherapy drug directly to a tumor. Because of the direct targeting, the new effort not only increases the effectiveness of paclitaxel, it also dramatically reduces devastating side effects such as hair loss, nausea, and nerve pain. CytImmune earlier this year asked David Kingston, a University Distinguished Professor of Chemistry with the Virginia Tech College of Science, to create a paclitaxel derivative that binds to gold-based nanoparticles while in the blood stream, only releasing the drug once it’s inside a cancerous tumor. Paclitaxel chemotherapy is widely used to treat breast, ovarian, lung, and colon cancer. 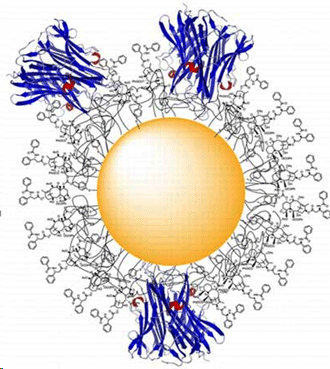 The new nanomedicine is composed of 27 nm gold nanoparticles, tumor necrosis factor alpha (TNFalpha), thiolated polyethylene glycol (PEG-thiol).
In other words, for now, delivery of a paclitaxel equals a shotgun with pellets. The blast of killing a tumor results in great collateral damage. Kingston and his team say their delivery method is like a finely tuned rifle, using CytImmune’s gold-based nanoparticles as the delivery bullet. The gold nanoparticles are decorated with both paclitaxel and tumor necrosis factor – a cell-signaling protein commonly called TNF. Gold nanoparticles are known to cling around cancerous tumors. TNF thus binds to the tumor blood vessel cells, ultimately killing them and reducing the high pressure inside the tumor, which prevents paclitaxel from reaching the cancer cells to kill them. Now, the slowly released paclitaxel that is bound to the gold nanoparticles can reach its targeted cancer cells to kill them. In early lab tests in treating mouse melanoma, a 2.5 milligram dose of paclitaxel delivered on Kingston’s gold nanoparticles vehicle was essentially as effective as a dose of 40 milligrams of paclitaxel by itself. The delivery method is expected to soon move toward clinical trial, said Kingston. Findings by Kingston and his team – including Jielu Zhao, a 2016 doctoral graduate in chemistry, now a chemist at Proctor and Gamble, and Shugeng Cao, a former post-doctorate researcher also in chemistry, now an associate professor at the University of Hawaii at Hilo — were recently published in the scientific journal Bioconjugate Chemistry ("Synthesis and Evaluation of Paclitaxel-Loaded Gold Nanoparticles for Tumor-Targeted Drug Delivery"). Zhao and Cao carried out the actual synthesis of the paclitaxel derivatives with the designed linkers to allow them to bond to the gold nanoparticles, with Kingston supervising. “This approach has the potential to be a game-changer in nanoparticle-based drug delivery systems,” said Kingston, “since it combines the power of drug targeting by tumor necrosis factor, with the advantages of nanoparticle delivery, including the low toxicity of nanoparticle drugs to normal, healthy tissue.” “By combining the tumor blood vessel destroying activity of TNF with the cancer killing effect of paclitaxel onto CytImmune’s tumor-targeted, ‘stealth’ gold nanoparticles, Dr. Kingston’s team and CytImmune’s team may have potentially created a new cancer drug that is far more effective and less toxic to the human body,” said Lawrence Tamarkin, chief executive officer at CytImmune. Work on the new drug was split between Virginia Tech’s main Blacksburg campus and CytImmune’s Rockville, Maryland, headquarters. Kingston has teamed with CytImmune in the past on tumor-targeting nanomedicine. Virginia Tech previously has used gold nanoparticles in unrelated anti-cancer research, including the Virginia-Maryland College of Veterinary Medicine which in experiments used gold nanoparticles to collect around tumors found inside a dog, and then utilized a non-ablative laser to target the gold nanoparticles, and thus the tumors. In essence, the veterinary approach killed cancer cells by heating them, versus Kingston’s approach directly targeting paclitaxel to tumors via the gold nanoparticles. Virginia Tech. Posted: Nov 14, 2016. |
|||||||||||||||||||||||||